 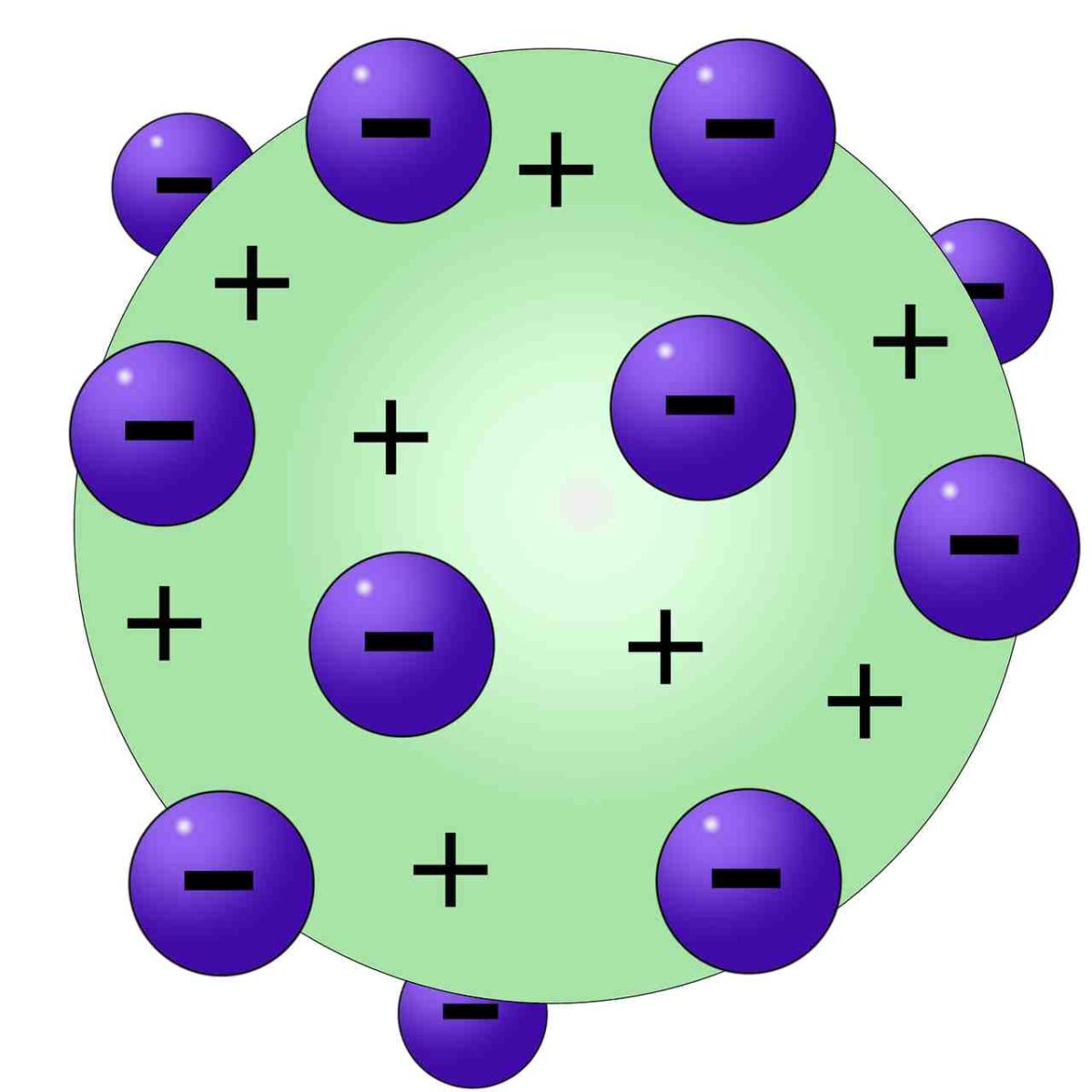
Thomson did still receive many honors during his lifetime, including being awarded the Nobel Prize in Physics in 1906 and a knighthood in 1908. This model became known as Thomson Atomic Model 1-4. However, this model of the atom soon gave way to a new model developed by New Zealander Ernest Rutherford (1871-1937) about five years later. He proposed that the atom was a sphere with charges embedded within it. Both subjects were transformed by the experiments of J. He concluded that all of the positive charge and the majority of the mass of the atom must be concentrated in a very small space in the atom's interior, which he called the nucleus.\): The "plum pudding" model (Credit: User: Fastfission/Wikimedia Commons Source: (opens in new window) License: Public Domain) Thomson, the Discovery of the Electron, and the Study of Atomic StructureOverviewLate in the nineteenth century physicists were working hard to understand the properties of electricity and the nature of matter. 
In contrast, the particles that were highly deflected must have experienced a tremendously powerful force within the atom. 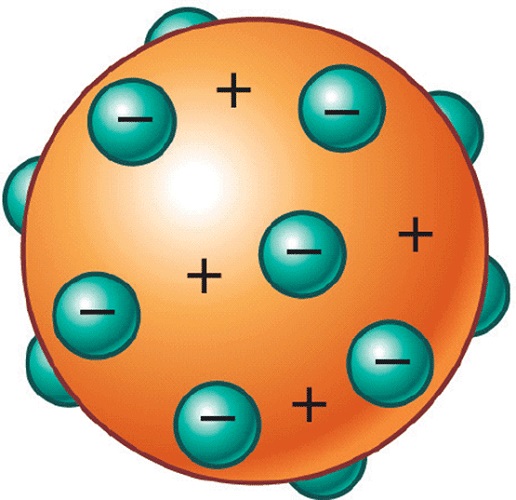
Because the vast majority of the alpha particles had passed through the gold, he reasoned that most of the atom was empty space. Rutherford needed to come up with an entirely new model of the atom in order to explain his results. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." Rutherford reasoned that if Thomsons model was correct then the mass of the atom. No prior knowledge had prepared them for this discovery. Rutherford tested Thomsons hypothesis by devising his gold foil experiment. Some were even redirected back toward the source. Thomson proposed a model of the atom in 1904 after doing a series of cathode tube experiments. Surprisingly, while most of the alpha particles were indeed not deflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. Following his discovery of the electron in 1897, British Physicist J.J. Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model, in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. JJ Thomson Proposed that an atom consists. Already in his 1913 trilogy, Bohr had sought to apply his theory to the understanding of the periodic table of elements. The electrons were assumed to be positioned in revolving circles around the atom in this model to be having a 'cloud' of positive charge. (B) According to the plum pudding model (top), all of the alpha particles should have passed through the gold foil with little or no deflection. Thomson, who invented the electron in the year 1897, suggested the atom's plum pudding model in 1904 which was for including the electron in the atomic model. The Thomson Model has negative partices (electrons) and a sphere of positive charge. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles. According to the postulates of Thomsons atomic model, an atom resembles a sphere of positive charge with electrons (negatively charged particles) present. Without any experimental evidence, he envisioned the atom as a diffuse sphere of positive charge with electrons randomly distributed inside. Make sure you have the correct idea firmly in mind. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
